|
Lide, (ed.), CRC Handbook of Chemistry and Physics 1999-2000 : A Ready-Reference Book of Chemical and Physical Data (CRC Handbook of Chemistry and Physics, CRC Press, Boca Raton, Florida, USA, 79th edition, 1998. I am grateful to Professor Pekka Pyykkö (University of Helsinki, Finland) who provided the nuclear quadrupole moment data in this and the following two references. Where given, data for certain radioactive nuclei are from this reference. Mason in Multinuclear NMR, Plenum Press, New York, USA, 1987. I am grateful to Professor Robin Harris (University of Durham, UK) who provided much of the NMR data, which are copyright 1996 IUPAC, adapted from his contribution contained within this reference. 5, John Wiley & Sons, Chichester, UK, 1996. 1: Moles to Mass Conversion with Elements. According to the periodic table, 1 mol of U has a mass of 238.0 g, so the mass of 2 mol is twice that, or 476.0 g. Harris in Encyclopedia of Nuclear Magnetic Resonance, D.M. The periodic table shows that the atomic mass (rounded to two decimal points) of Al is 26.98, so 1 mol of Al atoms has a mass of 26.98 g. Printed from Molar Mass, Molecular Weight and Elemental Composition Calculator. 
The atomic mass of tantalum is 180.9479 u. Magnetogyric ratio, γ (10 7 rad T ‑1 s -1) Molar mass calculator computes molar mass, molecular weight and elemental composition of any given compound. The typical isotope mass of tantalum is 181. Table of NMR-active nucleus propeties of hafnium For the purpose of this exercise, it will be assumed your cotton ball consists of simple glucose molecules. Note: Cellulose is a complex carbohydrate consisting of 3,000 or more glucose (C6H1005) units. Ĭommon reference compound: not available. Use the periodic table to determine the molar mass of this compound and record in Data Table 1. Now, compute the sum of products of number of atoms to the atomic weight: Molar mass (C 7 O 6 H) Count i Weight i. The smallest particles in a compound are molecules, and the smallest particles in elements are atoms. Then, lookup atomic weights for each element in periodic table: C: 12.0107, O: 15.9994, H: 1.00794. In chemistry, pure substances include all the elements on the periodic table, as well as all defined chemical compounds such as H 2 O. 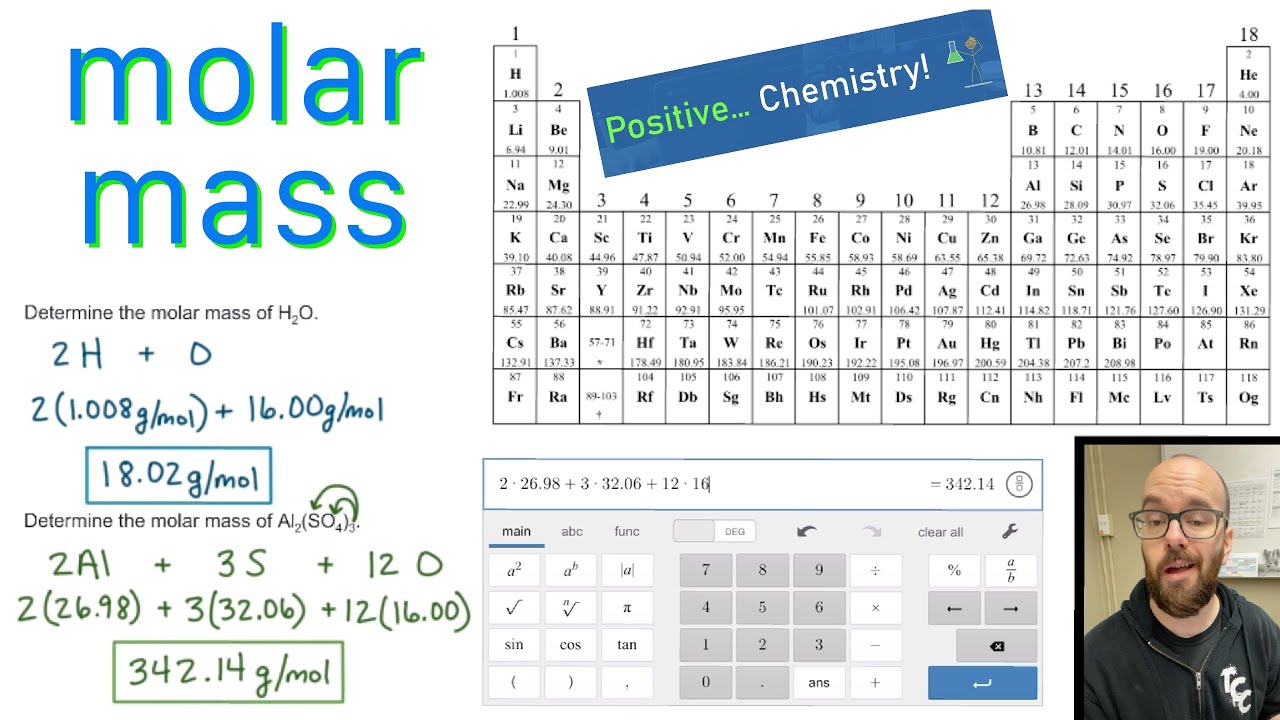
Kuchitsu in Quantities, Units and Symbols in Physical Chemistry, Blackwell Scientific Publications, Oxford, UK, 1988. First, compute the number of each atom in C 7 O 6 H: C: 7, O: 6, H: 1.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
